



|
Exploring the atmosphere |
|
Tianyi Fan’s Home Page |
|
Titan’s Methane Cycle |
|
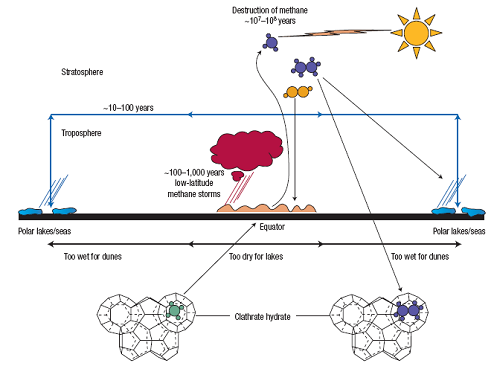
Hypothesis The short-term cycle of CH4 depletion by rain and recharge by evaporation are not the whole story. All the CH4 in Titan’s atmosphere will be photochemically destructed in 10 million years. If CH4 in Titans atmosphere all condense to the ground, there will be a kilometer-thick liquid layer. Unless tens of kilometers deep, the lakes are not plausible candidates for a long-term resupply reservoir of CH4. Then what is the source of long-term resupply? An extensive CH4 (or CH4/C2H6) equivalent to an aquifer system ― an ‘alkanofer’ ― might circulate liquids, especially beneath the lake features at high latitude. But the system would need to extend through tens of kilometers of the water ice crust of Titan (figure on right), and be densely packed. There is no evidence in the Cassini remote sensing data for or against such a system. A more plausible explanation is a source of CH4 from the deep interior. During its formation around Saturn, Titan could incorporated ammonia (NH3), hydrogen-bonded to water ice, and CH4, trapped in ice as clathrate hydrate if the condition is cold enough (cold-condition hypothesis). If the Saturnian nebula was too warm during Titan’s formation to allow CH4 to be trapped, CH4 could also enter the interior in the form of carbon grains, CO, CO2, and refractory organic material, which cloud have been later converted to CH4 by water-rock reactions with CH4 subsequently being stored as a clathrate hydrate in Titan and released over time (warm-condition hypothesis). |
Long-Term CH4 Cycle
|
|
In a geological time scale of 10 million years, the CH4 inventory in Titan’s atmosphere today will be photochemically destructed. There must be a long-term source of CH4 to replenish the loss. |
|
Titan’s interior structure |
|
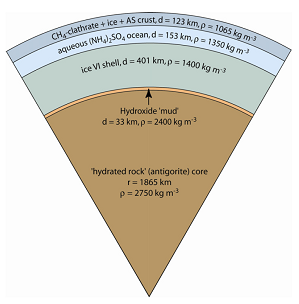
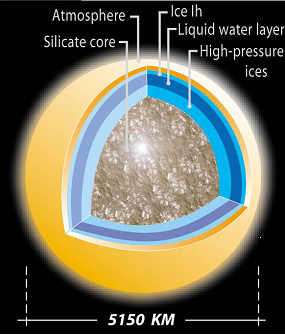
Titan’s interior (thin-crust model)[Tobie et al., 2005] |
|
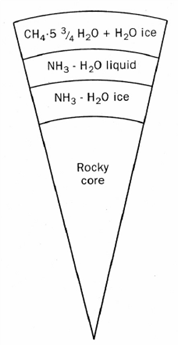
Titan’s interior (thick-crust model). [Fortes et al., 2007] |
|
Mechanism for CH4 outgassing How has interior CH4 been released to the surface? There are currently two theoretical models both based on observed phenomena on Titan. In either cases, substantial amounts of CH4 are stored in the interior― whether the gas arrive as CH4 or it was produced in the moon’s interior. The thin-crust model is based on the inference of Titan’s interior ocean. When constrained by the density of Titan, the equation of state of the relevant materials, and the available energy sources, Titan’s interior includes a liquid water ocean with perhaps a few percent of NH3, overlain by a crust (which today is mostly water ice but in the past was thin and largely composed of CH4 clathrate hydrate), a lower mantle of high-pressure phases of water ice, and a rock core (figure on right). The low thermal conductivity and high rigidity of the CH4 clathrate delay thickening of the crust, so that thermal events such as onset of convection in the core and later in the crust could result in CH4 outgassing to the surface, perhaps through the involvement of NH3-water cryovolcanism. The thick-crust model leach sulphur from the core which yields a thicker crust and a briny ocean containing dissolved ammonium sulphate ((NH4)2SO4). The presence of briny cryomagmas allows explosive dissociation of entrained methane (figure on right). This model is potentially more attractive if large amounts of CO2 ― which reacts with NH3 ― was present in Titan’s interior early in its history. In both models, long-period absence of volcanism is allowed so that Titan’s surface CH4 was fully consumed. Under such circumstances, a pure N2 could still supported surface temperature within 10 K as suggested by radiative balance studies. Surface deposits of C2H6 might have been frozen continuously or periodically leading to glacial erosion. |
|
Evidence There is evidence supporting the warm-condition hypothesis from Cassini VIMS instrument. CO2 has been detected on the surface and could well indicate outgassing of CO2 from interior, since photochemically produced CO2 is too limited to maintain the its surface value. Moreover, the lack of detection of noble gases, Xe and Kr by Huygens GCMS further support the CO2 origin hypothesis, as these gases should have been delivered together with CH4 if CH4 was captured directly (cold-condition hypothesis). However, it could not be rejected that these gases are still trapped with CH4 in the interior or there might be a sink of them that have removed them from Titan’s present-day atmosphere. |
|
The sink for C2H6 An outstanding problem in any evolutionary model is where C2H6 goes to? Persistent photolysis of CH4 over the age of solar system would have produce the equivalent of a global ocean of C2H6 of 100~200m. It is possible that the CH4 has been present in the atmosphere for only a fraction of the history. It is also possible that C2H6 is sequestered in clathrate hydrate in the crust, leading effectively to its permanent removal from the surface-atmosphere system. Recent observations suggest that C2H6 must been participating in further photochemical reactions, producing highly complex heavy hydrocarbon aerosols and removed from the atmosphere. However, quantitative analysis of how much C2H6 will be removed need further effort. References Lunine J.I. and Atreya S.K. The methane cycle on Titan. Nature Geoscience. Vol. 1. 2008 |
|
Titan’s short-term and long-term CH4 cycle. [Lunine and Atreya, 2008] |